Clinical Applications: Oncology
Neutrophil extracellular traps (NETs) in tumor-induced systemic ettects

ONCOLOGY
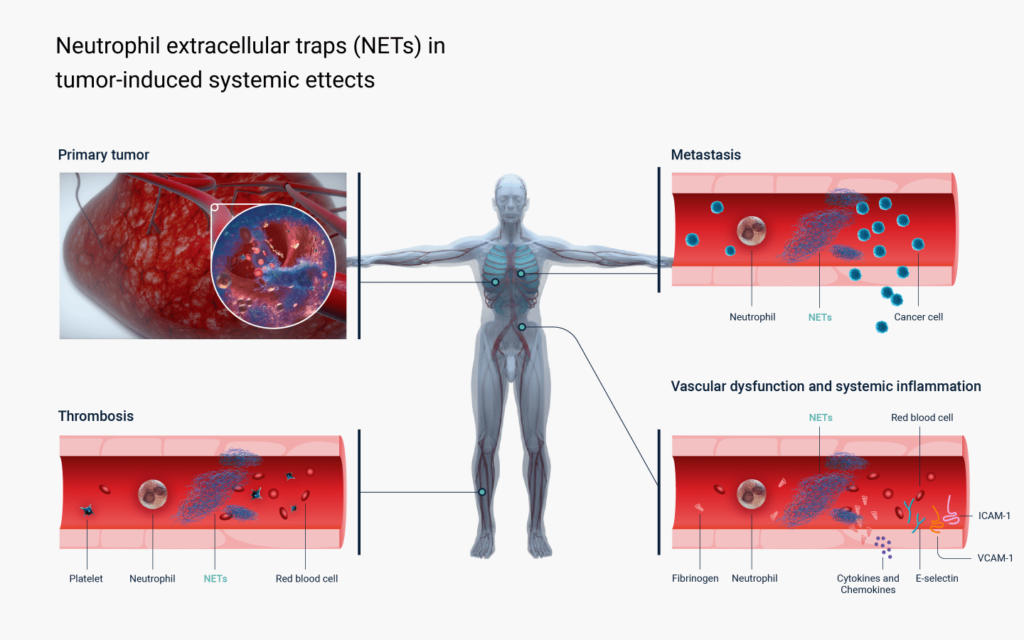
Elevated blood levels of NETs are a hallmark of cancer. It has been shown that cancer cells provoke the innate immune system to produce more NETs in order to promote their own growth, invasion and metastasis.
Recent studies have confirmed that NETs have the ability to direct multiple areas associated with cancer pathology including cell genomic instability, hyper-mutation, migration, invasion, metastasis formation, vascularization and immune evasion. In patients with advanced disease, NETs facilitate the development of thrombosis, coagulopathies, hyperinflammatory state, systemic immunosuppression and organ failure. In patients receiving anticancer treatment NETs release promotes the spread of drug resistance and dose-limiting systemic toxicity.
Multiple animal studies have confirmed that pharmacological prevention of NET release, or its enzymatic degradation, produce substantial therapeutic benefit in cancer models. These are exhibited by inhibition of tumour growth and metastasis, boosting efficacy of chemotherapy and immunotherapy as well as the correction of coagulation and organ dysfunction.
In contrast to any potential pharmacological interventions, removal of NETs with NucleoCapture allows removal of NETs from blood without compromising the defensive function of the innate immune system.
We believe that the removal of NETs using NucleoCapture therapeutic apheresis might represent a powerful and safe adjunctive option for patients receiving anticancer treatment.
